
Zheng Chen, PhD
- Professor
- Vice Chair of Research
Areas of Interest
Research Interests
Circadian clocks, Small molecule modulators , Metabolic disease, Aging, Nutrition and Diets, Autophagy, Microbiome
Small-molecule probes for chronobiology and medicine
The circadian clock is our intrinsic biological timer orchestrating essential cellular and physiological processes. A major research effort in my lab focuses on small molecules capable of enhancing the amplitude, or robustness, of the circadian clock. We hypothesize that such clock-enhancing small molecules (dubbed CEMs) will improve timing and therefore performance of fundamental physiological pathways, which can be exploited for therapeutic gains in aging and chronic diseases such as metabolic disease known to suffer dampened circadian rhythms.
My lab previously identified CEMs (PNAS 2012; Cell Mol Life Sci 2013) via high-throughput chemical screening and showed that these compounds can potently enhance peripheral and/or central circadian clocks. In a recent breakthrough study, we showed that a naturally occurring flavonoid enriched in citrus peels, called Nobiletin, is a robust CEM, and displays potent clock-dependent protection against metabolic diseases in both diet- and mutation-induced mouse models (Cell Metabolism 2016; Nutr Metab 2015). Importantly, we identified the nuclear receptors RORs as the direct protein target of Nobiletin (see Figure). These studies laid an excellent foundation for our continuing investigation of circadian regulation of energy homeostasis. Extending beyond metabolic studies, we are also gaining important insights into the roles of Nobiletin and RORs in healthy aging and other clock-related diseases. A long-term goal of this project is to develop novel Nobiletin derivatives for translational and clinical applications.
We have also elucidated novel regulatory mechanisms of metabolic homeostasis. Following a serendipitous finding where a partial Clock-deficient mouse mutant surprisingly showed compensatory metabolic enhancement, our in vitro studies uncovered a previously unknown mechanism involving autophagy-dependent turnover of the central clock transcription factor BMAL1 (Jeong et al., Sci Rep 2015). Another emerging topic in our lab is the microbial regulatory mechanism for energy balance. Combining microbiome sequencing, metabolomics and fecal microbiota transplantation, we showed that dietary fiber causes transmissible microbial and metabolomic remodeling, leading to improved glucose homeostasis in diabetic mice (He et al., Sci Rep 2015). These exciting on-going projects are expected to synergize with small-molecule studies, and may lead to innovative therapeutic regimens.
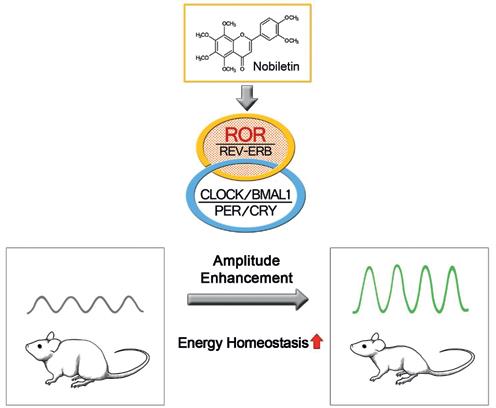
Nobiletin as a clock enhancing molecule (CEM). Nobiletin directly targets the clock oscillator to enhance circadian amplitude, thereby promoting energy homeostasis and protecting against metabolic syndrome
Selected Publications
He B, Nohara K, Park N, YS, Guillory B, Zhao Z, Garcia JM, Koike N, Lee CC, Takahashi JS, Yoo SH, Chen Z. 2016. The small molecule Nobiletin targets the molecular oscillator to enhance circadian rhythms and protect against metabolic syndrome. Cell Metab. 23: 610-21.
Chen Z, Yoo SH, Park YS, Kim KH, Wei S, Buhr E, Ye ZY, Pan HL, Takahashi JS. 2012. Identification of diverse modulators of central and peripheral circadian clocks by high-throughput chemical screening. Proc Natl Acad Sci U S A. 109(1):101-6.
Nohara K, Shin Y, Park N, Jeong K, He B, Koike N, Yoo SH, Chen Z . 2015. Ammonia- lowering activities and carbamoyl phosphate synthetase 1 (Cps1) induction mechanism of a natural flavonoid. Nutr Metab (Lond). 12:23. eCollection 2015.
Education and Training
BS
Biological Sciences, Tsinghua University
PhD
Molecular Biology and Biochemistry, Columbia University
Postdoctoral Fellow
Biochemistry, University of Texas Southwestern Medical School
Graduate Program Affiliations
Molecular and Translational Biology