Biography
Dr. Krachler joined the faculty of the Department of Microbiology and Molecular Genetics at UTHealth Houston’s McGovern Medical School as an Associate Professor in September 2016. She completed her undergraduate and masters degree in Chemical Engineering at the Vienna University of Technology, Austria, before training for a Ph.D. in Biochemistry with Colin Kleanthous at the University of York in the UK. After joining Kim Orth’s lab at UT Southwestern Medical Center as a postdoctoral fellow, she moved to the University of Birmingham as an EMBO Fellow and Birmingham Fellow in 2012, where she first established her independent laboratory.
Dr Krachler’s research on bacteria-host interactions has been recognized by several awards, including a Texas Rising STAR award (2016), the Biochemical Society’s Early Career Research Award for Biological Systems (2015) and a Founder’s Award (2015).
Education
- Postdoctoral Fellow
- UT Southwestern Medical Center
- Ph.D.
- University of York, United Kingdom
Areas of Interest
Research Interests
We take a highly interdisciplinary approach to study information exchange at the bacteria-host interface. The main questions driving this work are “How do bacteria perceive the host and react to switch on colonization and virulence mechanisms? And how, in turn, does the biophysical process of bacterial adhesion affect the host in a way that facilitates colonization and infection?” While deepening our understanding of the basic biology of bacteria-host interactions, we concurrently work on translating our discoveries into clinically relevant applications, focusing on novel therapeutics to combat drug-resistant bacterial infections. Some of the projects we are currently pursuing are:
1. Developing adhesion inhibitors to counteract bacterial infections
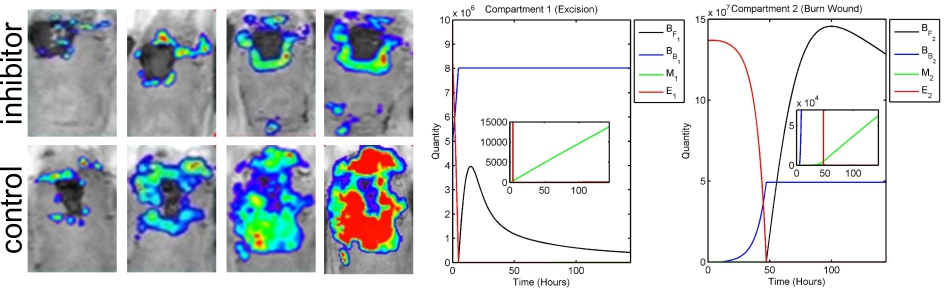
Antimicrobial resistance is a growing threat to human health, and many bacterial infections are becoming difficult to treat as a result. We are developing novel anti-infectives, which inhibit infections by blocking a pathogen’s ability to stick to tissue and wound surfaces and facilitate their removal from the host. This work involves biochemistry and biological chemistry for the design and characterization of such inhibitors. We also collaborate with mathematicians and clinicians, to predict how modifications to the inhibitors will improve their potency, and then evaluate their use against drug-resistant wound- and surgical site infections.
Publications
Mahmoud RY, Stones DH, Li W, Emara M, El-Domany RA, Wang D, Wang Y, Krachler AM, Yu J. (2016) The Multivalent Adhesion Molecule SSO1327 plays a key role in Shigella sonnei pathogenesis. Mol Microbiol. 99(4):658-73. PMID: 26481305
Alsharif G, Ahmad S, Islam MS, Shah R, Busby SJ, Krachler AM. (2015) Host attachment and fluid shear are integrated into a mechanical signal regulating virulence in Escherichia coli O157:H7. Proc Natl Acad Sci U S A. 112(17):5503-8. PMID: 25870295
Ternent L, Dyson RJ, Krachler AM, Jabbari S. (2015) Bacterial fitness shapes the population dynamics of antibiotic-resistant and -susceptible bacteria in a model of combined antibiotic and anti-virulence treatment. J Theor Biol. 7;372:1-11. PMID: 25701634
Lim J, Stones DH, Hawley CA, Watson CA, Krachler AM. (2014) Multivalent adhesion molecule 7 clusters act as signaling platform for host cellular GTPase activation and facilitate epithelial barrier dysfunction. PLoS Pathog. 10(9):e1004421. PMID: 25255250
Krachler AM, Ham H, Orth K.(2011) Outer membrane adhesion factor multivalent adhesion molecule 7 initiates host cell binding during infection by gram-negative pathogens. Proc Natl Acad Sci U S A. 108(28):11614-9. PMID: 2170922