Research
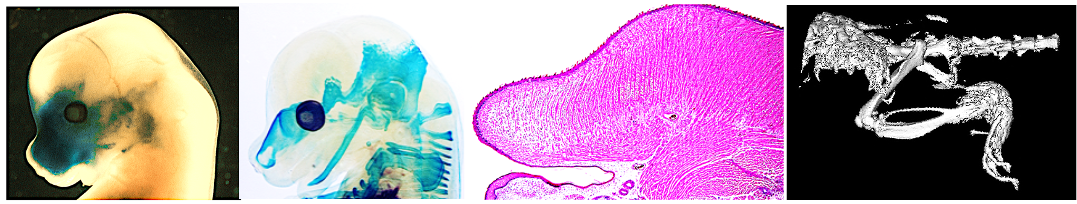
The abnormal growth of facial bone and cartilage frequently leads to congenital craniofacial malformations that result in around 30% of all congenital diseases. Craniofacial skeletal abnormalities are largely attributed to defects in the formation, migration and differentiation of neural crest cells (NCCs), which are critical multipotent cell population generates a diverse array of craniofacial tissues. However, the mechanistic details of how the NCCs contribute to the subsequent induction of the skeletal tissues remain elusive. We employ multiple approaches including mouse genetics and sophisticated imaging techniques to define the etiology for craniofacial skeletal defects.
The role of ciliary protein for bone homeostasis in neural crest cells: The primary cilium is a cellular organelle that coordinates signaling pathways critical for cell survival and homeostasis. Intraflagellar transport (IFT) plays a pivotal role in assembling primary cilia. Disruption and/or dysfunction of IFT components can cause multiple diseases, including skeletal dysplasias. To date, their role to assemble cilia, which transduce a wide range of developmental signals (termed here as “ciliary-dependent role”), has been well appreciated. Additionally, recent studies conducted by ourselves and others have started to highlight the critical role of IFT proteins, which can have functions other than assembling cilia (termed here as “ciliary-independent role”). Studying the combination of both ciliary-dependent and ciliary-independent roles of IFT proteins may shed light on the complete etiology of craniofacial bone defects.
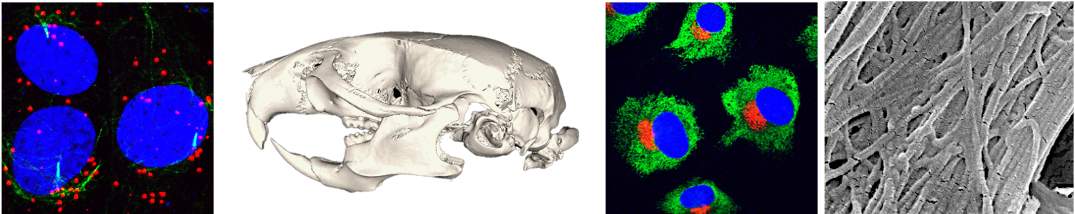
The role of DNA damage response in craniofacial development: Appropriate cellular responses to DNA damage are critically important for normal embryonic development. If damaged DNA is not repaired, cells lose their genomic integrity, which frequently results in uncontrollable cell proliferation and cell death. Thus, multiple birth defects are characterized by defective DNA damage response (DDR), which is an important cellular pathway that repairs damaged DNA. However, it remains unclear how and whether defective DDR actively participates in the etiology of craniofacial defects. If we can answer this question, we may be able to identify a novel mechanism that explains why craniofacial abnormalities are very common among congenital birth defects and leads to phenotypic variability among affected individuals.
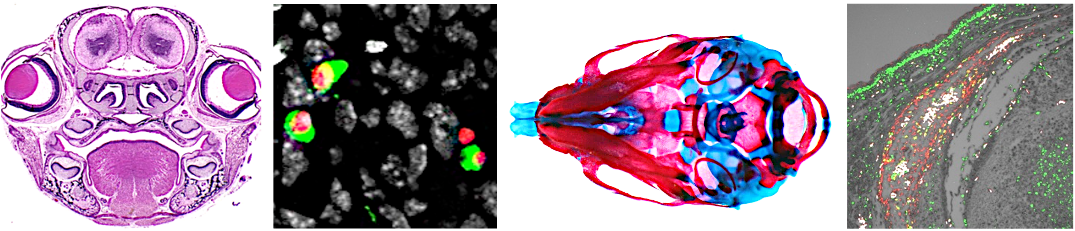
The role of growth factor signaling in craniofacial development: Bone morphogenetic proteins (BMPs) were identified by their ability to develop bone and BMP signaling is recognized as one of the important factors for inducing NCCs. However, it remains elusive how BMP signaling controls the skeletogenic fate and lineages in NCCs during craniofacial skeletal development. Our studies suggest that precise range of BMP signaling, i.e., not too-low or -high BMP signaling, is required for proper craniofacial skeletal development. In humans, activation of BMP signaling through mutated ACVR1 receptor causes ectopic bone formation seen in fibrodysplasia ossificans progressiva (FOP) . To date, it remains unclear how a gain-of-function mutation in ACVR1 develops the ectopic bones in the craniofacial region. Our study may provide molecular insight to explain how an excess amount of BMP signaling regulates the chondrogenic fate in NCCs and leads to craniofacial skeletal malformations in FOP patients.